PDF Publication Title:
Text from PDF Page: 190
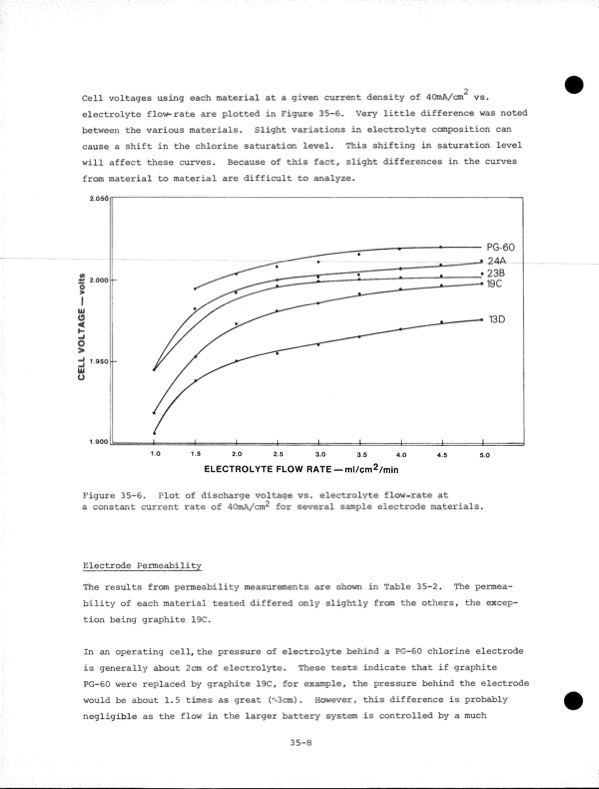
Cell voltages using each material at a given current density of 4OmA/cm vs. electrolyte flow-rate are plotted in Figure 35-6. Very little difference was noted between the various materials. Slight variations in electrolyte composition can cause a shift in the chlorine saturation level. This shifting in saturation level will affect these curves. Because of this fact, slight differences in the curves from material to material are difficult to analyze. 2.050 2.000 PG-60 24A 23B 19C 1.900 ELECTROLYTE FLOW RATE — ml/cm2/min Figure 35-6. Plot of discharge voltage vs. electrolyte flow-rate at a constant current rate of 40mA/cm^ for several sample electrode materials. Electrode Permeability The results from permeability measurements are shown in Table 35-2. The permea bility of each material tested differed only slightly from the others, the excep tion being graphite 19C. In an operating cell, the pressure of electrolyte behind a PG-60 chlorine electrode is generally about 2cm of electrolyte. These tests indicate that if graphite PG-60 were replaced by graphite 19C, for example, the pressure behind the electrode would be about 1.5 times as great ('x-Scm) . However, this difference is probably negligible as the flow in the larger battery system is controlled by a much 35-8 CELL VOLTAGEPDF Image | Development of the Zinc-Chlorine Battery for Utility
PDF Search Title:
Development of the Zinc-Chlorine Battery for UtilityOriginal File Name Searched:
6302789.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
CONTACT TEL: 608-238-6001 Email: greg@salgenx.com (Standard Web Page)