
PDF Publication Title:
Text from PDF Page: 214
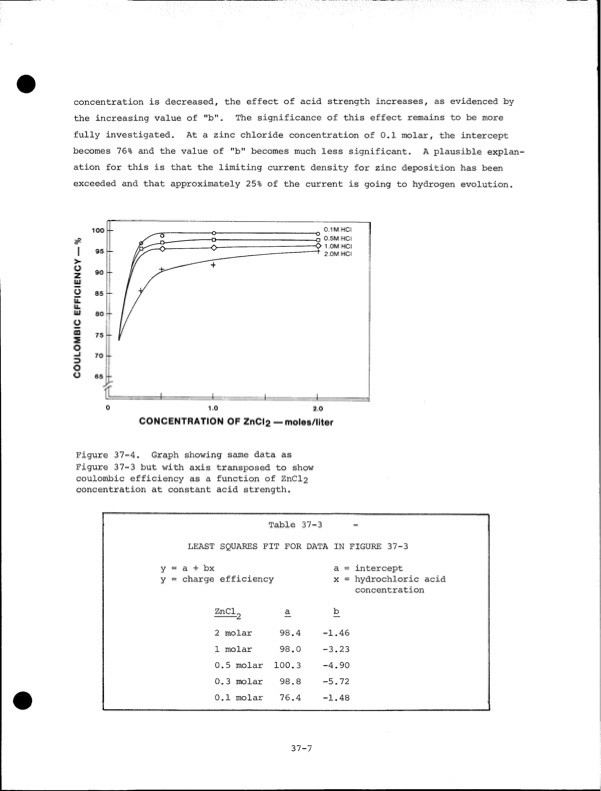
concentration is decreased, the effect of acid strength increases, as evidenced by the increasing value of "b". The significance of this effect remains to be more fully investigated. At a zinc chloride concentration of 0.1 molar, the intercept becomes 76% and the value of "b" becomes much less significant. A plausible explan ation for this is that the limiting current density for zinc deposition has been exceeded and that approximately 25% of the current is going to hydrogen evolution. 0.1 M HCI 2 0.5M HCI <> 1.0M HCI 2.0M HCI 0 1.0 2.0 CONCENTRATION OF ZnCl2 — moles/liter Figure 37-4. Graph showing same data as Figure 37-3 but with axis transposed to show coulombic efficiency as a function of ZnCl2 concentration at constant acid strength. Table 37-3 - LEAST SQUARES FIT FOR DATA IN FIGURE 37-3 y = a + bx y = charge efficiency ZnCl„ a 2 molar 98.4 1 molar 98.0 0.5 molar 100.3 0.3 molar 98.8 0.1 molar 76.4 a = intercept x = hydrochloric acid concentration 37-7 b -1.46 -3.23 -4.90 -5.72 -1.48PDF Image | Development of the Zinc-Chlorine Battery for Utility
PDF Search Title:
Development of the Zinc-Chlorine Battery for UtilityOriginal File Name Searched:
6302789.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
| CONTACT TEL: 608-238-6001 Email: greg@salgenx.com | RSS | AMP |