
PDF Publication Title:
Text from PDF Page: 231
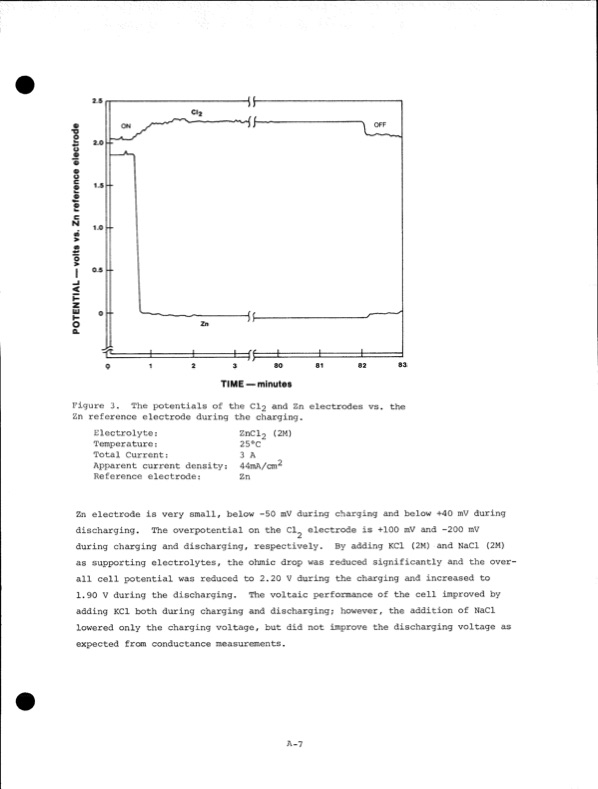
TIME — minutes Figure 3, The potentials of the CI2 and Zn electrodes vs. the Zn reference electrode during the charging. Electrolyte: Temperature: Total Current: Apparent current density: Reference electrode: ZnCl2 (2M) 25°C 3 A 44mA/cm2 Zn Zn electrode is very small, below -50 mV during charging and below +40 mV during discharging. The overpotential on the Cl^ electrode is +100 mV and -200 mV during charging and discharging, respectively. By adding KC1 (2M) and NaCl (2M) as supporting electrolytes, the ohmic drop was reduced significantly and the over all cell potential was reduced to 2.20 V during the charging and increased to 1.90 V during the discharging. The voltaic performance of the cell improved by adding KC1 both during charging and discharging; however, the addition of NaCl lowered only the charging voltage, but did not improve the discharging voltage as expected from conductance measurements. A-7PDF Image | Development of the Zinc-Chlorine Battery for Utility
PDF Search Title:
Development of the Zinc-Chlorine Battery for UtilityOriginal File Name Searched:
6302789.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
| CONTACT TEL: 608-238-6001 Email: greg@salgenx.com | RSS | AMP |