
PDF Publication Title:
Text from PDF Page: 067
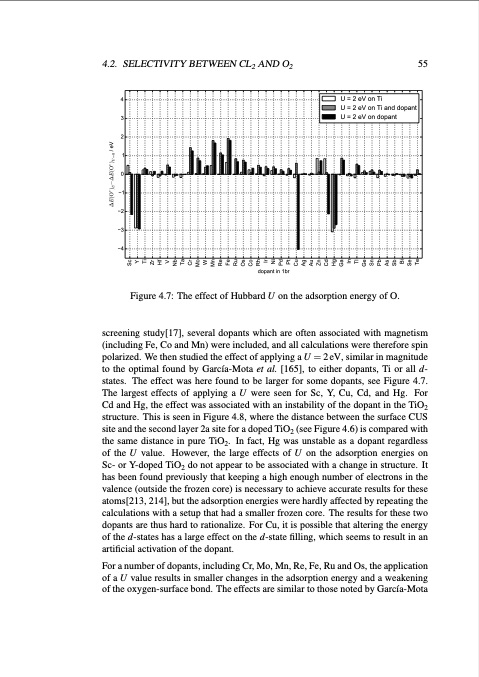
4.2. SELECTIVITY BETWEEN CL2 AND O2 55 U = 2 eV on Ti U = 2 eV on Ti and dopant U = 2 eV on dopant 4 3 2 1 0 1 2 3 4 dopant in 1br Figure 4.7: The effect of Hubbard U on the adsorption energy of O. screening study[17], several dopants which are often associated with magnetism (including Fe, Co and Mn) were included, and all calculations were therefore spin polarized. We then studied the effect of applying a U = 2 eV, similar in magnitude to the optimal found by García-Mota et al. [165], to either dopants, Ti or all d- states. The effect was here found to be larger for some dopants, see Figure 4.7. The largest effects of applying a U were seen for Sc, Y, Cu, Cd, and Hg. For Cd and Hg, the effect was associated with an instability of the dopant in the TiO2 structure. This is seen in Figure 4.8, where the distance between the surface CUS site and the second layer 2a site for a doped TiO2 (see Figure 4.6) is compared with the same distance in pure TiO2. In fact, Hg was unstable as a dopant regardless of the U value. However, the large effects of U on the adsorption energies on Sc- or Y-doped TiO2 do not appear to be associated with a change in structure. It has been found previously that keeping a high enough number of electrons in the valence (outside the frozen core) is necessary to achieve accurate results for these atoms[213, 214], but the adsorption energies were hardly affected by repeating the calculations with a setup that had a smaller frozen core. The results for these two dopants are thus hard to rationalize. For Cu, it is possible that altering the energy of the d-states has a large effect on the d-state filling, which seems to result in an artificial activation of the dopant. For a number of dopants, including Cr, Mo, Mn, Re, Fe, Ru and Os, the application of a U value results in smaller changes in the adsorption energy and a weakening of the oxygen-surface bond. The effects are similar to those noted by García-Mota Sc Y Ti Zr Hf V Nb Ta Cr Mo W Mn Re Fe Ru Os Co Rh Ir Ni Pd Pt Cu Ag Au Zn Cd Hg Ga In Tl Ge Sn Pb As Sb Bi Se Te ∆E(Oc )U−∆E(Oc )U =0 / eVPDF Image | Studies of Electrode Processes in Industrial Electrosynthesis
PDF Search Title:
Studies of Electrode Processes in Industrial ElectrosynthesisOriginal File Name Searched:
electrosynthesis.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
| CONTACT TEL: 608-238-6001 Email: greg@salgenx.com | RSS | AMP |