PDF Publication Title:
Text from PDF Page: 233
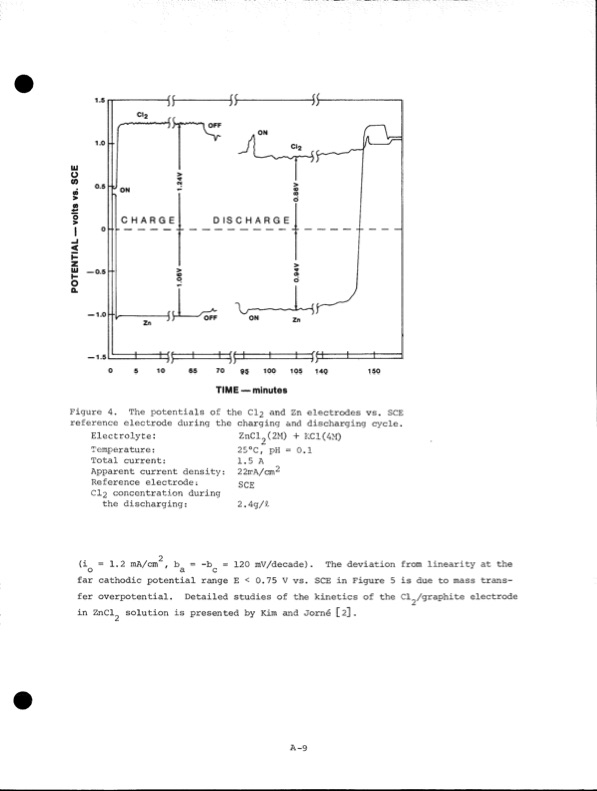
— 0.5 — 1.0 — 1.5 CHARGE DISCHARGE Electrolyte: Temperature: Total current: Apparent current density: Reference electrode; CI2 concentration during the discharging: ZnCl?(2M) + KC1(4M) 250C,~ pH = 0.1 1.5 A 22irA/cm^ SCE 2.4g/£ TIME — minutes Figure 4. The potentials of the Cl? and Zn electrodes vs. SCE reference electrode during the charging and discharging cycle. 2 (iQ =1.2 mA/cm , = -b^ = 120 mV/decade). The deviation from linearity at the far cathodic potential range E < 0.75 V vs. SCE in Figure 5 is due to mass trans fer overpotential. Detailed studies of the kinetics of the Cl^/graphite electrode in ZnCl^ solution is presented by Kim and Jorne [2]. A-9 POTENTIAL — volts vs. SCEPDF Image | Development of the Zinc-Chlorine Battery for Utility
PDF Search Title:
Development of the Zinc-Chlorine Battery for UtilityOriginal File Name Searched:
6302789.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
CONTACT TEL: 608-238-6001 Email: greg@salgenx.com (Standard Web Page)