
PDF Publication Title:
Text from PDF Page: 062
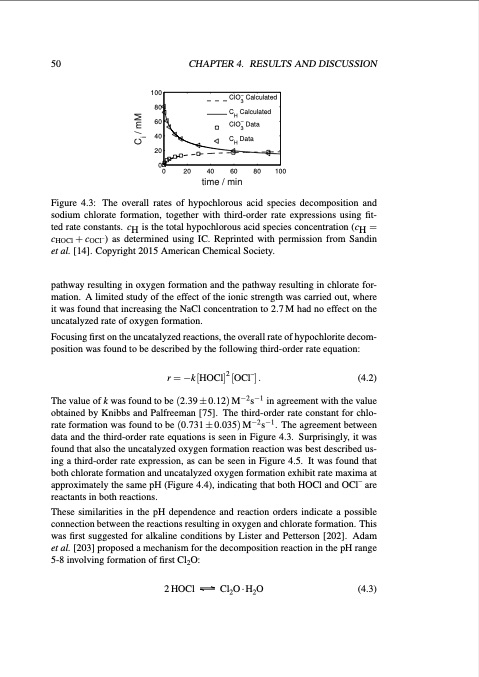
50 CHAPTER4. RESULTSANDDISCUSSION 100 80 60 40 20 0 0 20 40 60 80 100 time / min − ClO Calculated 3 C Calculated H − ClO Data 3 C Data H Figure 4.3: The overall rates of hypochlorous acid species decomposition and sodium chlorate formation, together with third-order rate expressions using fit- ted rate constants. cH is the total hypochlorous acid species concentration (cH = cHOCl +cOCl–) as determined using IC. Reprinted with permission from Sandin et al. [14]. Copyright 2015 American Chemical Society. pathway resulting in oxygen formation and the pathway resulting in chlorate for- mation. A limited study of the effect of the ionic strength was carried out, where it was found that increasing the NaCl concentration to 2.7 M had no effect on the uncatalyzed rate of oxygen formation. Focusing first on the uncatalyzed reactions, the overall rate of hypochlorite decom- position was found to be described by the following third-order rate equation: r = −k [HOCl]2 [OCl–] . (4.2) The value of k was found to be (2.39 ± 0.12) M−2 s−1 in agreement with the value obtained by Knibbs and Palfreeman [75]. The third-order rate constant for chlo- rate formation was found to be (0.731±0.035)M−2s−1. The agreement between data and the third-order rate equations is seen in Figure 4.3. Surprisingly, it was found that also the uncatalyzed oxygen formation reaction was best described us- ing a third-order rate expression, as can be seen in Figure 4.5. It was found that both chlorate formation and uncatalyzed oxygen formation exhibit rate maxima at approximately the same pH (Figure 4.4), indicating that both HOCl and OCl– are reactants in both reactions. These similarities in the pH dependence and reaction orders indicate a possible connection between the reactions resulting in oxygen and chlorate formation. This was first suggested for alkaline conditions by Lister and Petterson [202]. Adam et al. [203] proposed a mechanism for the decomposition reaction in the pH range 5-8 involving formation of first Cl2O: 2 HOCl Cl2O · H2O (4.3) C / mM iPDF Image | Studies of Electrode Processes in Industrial Electrosynthesis
PDF Search Title:
Studies of Electrode Processes in Industrial ElectrosynthesisOriginal File Name Searched:
electrosynthesis.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
| CONTACT TEL: 608-238-6001 Email: greg@salgenx.com | RSS | AMP |