PDF Publication Title:
Text from PDF Page: 086
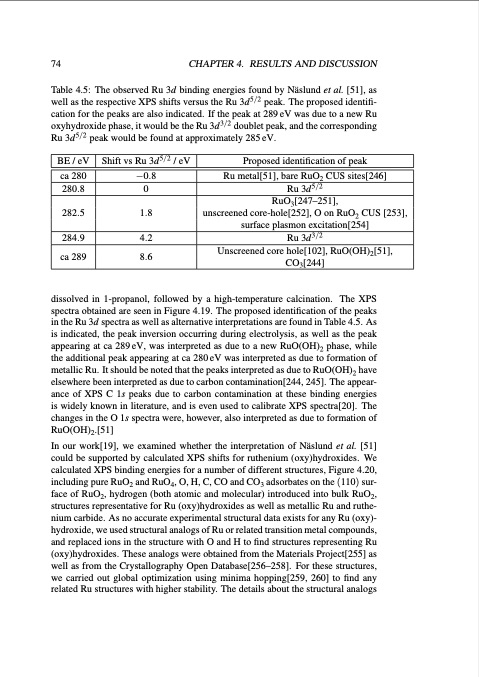
74 CHAPTER4. RESULTSANDDISCUSSION Table 4.5: The observed Ru 3d binding energies found by Näslund et al. [51], as well as the respective XPS shifts versus the Ru 3d5/2 peak. The proposed identifi- cation for the peaks are also indicated. If the peak at 289 eV was due to a new Ru oxyhydroxide phase, it would be the Ru 3d3/2 doublet peak, and the corresponding Ru 3d5/2 peak would be found at approximately 285 eV. BE / eV ca 280 280.8 282.5 284.9 ca 289 Shift vs Ru 3d5/2 / eV −0.8 0 1.8 4.2 8.6 Proposed identification of peak Ru metal[51], bare RuO2 CUS sites[246] Ru 3d5/2 RuO3[247–251], unscreened core-hole[252], O on RuO2 CUS [253], surface plasmon excitation[254] Ru 3d3/2 Unscreened core hole[102], RuO(OH)2[51], CO3[244] dissolved in 1-propanol, followed by a high-temperature calcination. The XPS spectra obtained are seen in Figure 4.19. The proposed identification of the peaks in the Ru 3d spectra as well as alternative interpretations are found in Table 4.5. As is indicated, the peak inversion occurring during electrolysis, as well as the peak appearing at ca 289 eV, was interpreted as due to a new RuO(OH)2 phase, while the additional peak appearing at ca 280 eV was interpreted as due to formation of metallic Ru. It should be noted that the peaks interpreted as due to RuO(OH)2 have elsewhere been interpreted as due to carbon contamination[244, 245]. The appear- ance of XPS C 1s peaks due to carbon contamination at these binding energies is widely known in literature, and is even used to calibrate XPS spectra[20]. The changes in the O 1s spectra were, however, also interpreted as due to formation of RuO(OH)2.[51] In our work[19], we examined whether the interpretation of Näslund et al. [51] could be supported by calculated XPS shifts for ruthenium (oxy)hydroxides. We calculated XPS binding energies for a number of different structures, Figure 4.20, including pure RuO2 and RuO4, O, H, C, CO and CO3 adsorbates on the (110) sur- face of RuO2, hydrogen (both atomic and molecular) introduced into bulk RuO2, structures representative for Ru (oxy)hydroxides as well as metallic Ru and ruthe- nium carbide. As no accurate experimental structural data exists for any Ru (oxy)- hydroxide, we used structural analogs of Ru or related transition metal compounds, and replaced ions in the structure with O and H to find structures representing Ru (oxy)hydroxides. These analogs were obtained from the Materials Project[255] as well as from the Crystallography Open Database[256–258]. For these structures, we carried out global optimization using minima hopping[259, 260] to find any related Ru structures with higher stability. The details about the structural analogsPDF Image | Studies of Electrode Processes in Industrial Electrosynthesis
PDF Search Title:
Studies of Electrode Processes in Industrial ElectrosynthesisOriginal File Name Searched:
electrosynthesis.pdfDIY PDF Search: Google It | Yahoo | Bing
Salgenx Redox Flow Battery Technology: Power up your energy storage game with Salgenx Salt Water Battery. With its advanced technology, the flow battery provides reliable, scalable, and sustainable energy storage for utility-scale projects. Upgrade to a Salgenx flow battery today and take control of your energy future.
CONTACT TEL: 608-238-6001 Email: greg@salgenx.com (Standard Web Page)